The Reaction of Butane With Oxygen Is Called
Gaseous butane CH3CHCH reacts with gaseous oxygen gas 02 to produce gaseous carbon dioxide CO and gaseous water HO. 2C4h10 g13O2 g-10H2O g8CO2 g Calculate the mass of water produced when 177 grams of butane reacts with excessive oxygen.

Complete Combustion Of Butane C4h10 Balanced Equation Youtube
Olo Ar Explanation Check 2022 McGraw Hill LLC.
. In any balanced chemical equation the number of each type of atom on both sides of the equation is ________. Organic compounds with carbon-carbon double or triple bonds are classified 22 A unsaturated compounds B saturated compounds C dilute solutions D concentrated solutions E substituted compounds as 23. The reaction of butane with oxygen is called 21 A substitution B combustion C neutralization D addition E titration 22.
What is the theoretical yield of carbon dioxide formed from the reaction of 151 g of butane and 330 g of oxygen gas. The conversion of cyclohexene to cyclohexane requires the reagents H2 and Pt. Be sure your answer has the correct number of significant digits in it.
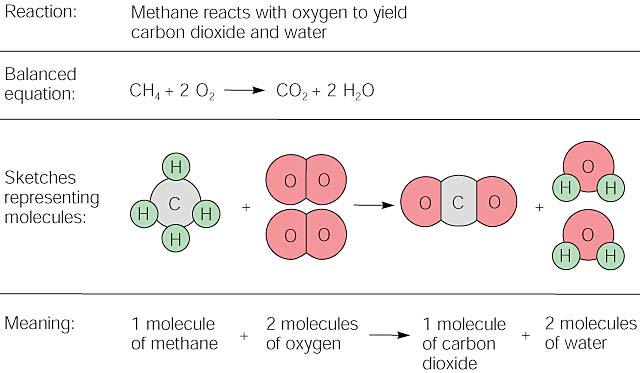
When butane CH10 reacts with oxygen carbon dioxide and water are produced. 2C4H10 g 1302 g 8CO2 g 10H2O g If 6 moles of butane CH10 react The reaction consumes moles of oxygen. 2C4H10g 13O2g 8CO2g 10H2O g Answer link.
Calculate the mass of butane needed to produce 716 of carbon dioxide. Gaseous butane CHCH CH reacts with gaseous oxygen gas o to produce gaseous carbon dioxide CO and gaseous water HO. The reaction of butane with oxygen is called a.
The balanced equation for this reaction is. Gaseous butane CH3CHCH reacts with gaseous oxygen gas 02 to produce gaseous carbon dioxide CO2 and gaseous water HO. When hydrogen is added to CH3CH2CHCHCH3.
The reaction of butane with oxygen is called addition. The reaction is a n. The reaction of butane with oxygen is called Asubstitution.
Be sure your answer has the correct number of significant digits in it. Your email address will not be published. The reaction produces moles of carbon dioxide and moles of water.
Below is a hydrocarbon combustion animation showing the net reaction that occurs when butane combines with oxygen. Butane releases its chemical energy by undergoing hydrocarbon combustion. Tertiary butyl hydroperoxide and tertiary butyl alcohol are prepared from isobutane and oxygen in a vertical reactor by sparging a mixture of isobutane with oxygen to the bottom of the reactor charging a reaction mixture recycle stream to the reactor above the sparge point centrally charging a downwardly flowing stream of cooled fresh isobutane to the top of the reactor to.
The reaction of hydrogen fluoride with ethene would be an example of. The reaction of butane with oxygen is called ________. Leave a Reply Cancel reply.
What is the theoretical yield of water formed from the reaction of 12 g of butane and 33 g of oxygen gas. The reaction of butane with oxygen is called. The chemical formula of butane is C4H10.
What is the 2 theoretical yield of carbon dioxide formed from the reaction of 29 g of butane and 40 g of oxygen gas. The reaction of butane with oxygen is called Addition Substitution Titration Neutralization Combustion. The combustion of butane is a reaction between butane and oxygen gas that produces carbon dioxide gas and water.
2 C 4 H 10 13 O 2 8 CO 2 10 H 2 O Heat Energy. Round your answer to 2 significant figures. Amines contain the element ________.
Butane C4H10 reacts with oxygen O2 to form water H2O and carbon dioxide CO2 as shown in the following chemical equation. Required fields are marked Comment Name.

How To Balance C4h10 O2 Co2 H2o Butane Combustion Reaction Youtube

Solved 11 The Reaction Of Butane With Oxygen Is Called Chegg Com

Solved The Reaction Of Butane With Oxygen Is Called Addition Chegg Com
Comments
Post a Comment